Abstract
Introduction: We and others have reported the role of cytokines and the tumor microenvironment on the development of severe grade >3 toxicities and CAR T resistance in recipients of CD19 targeted therapy. However, these analyses are not readily available outside of research settings. Early intervention and prophylactic strategies with cytokine blocking agents and/or steroids to mitigate severe toxicity have been explored. In a recent analysis of cohort 6 of the ZUMA-1 trial, investigators reported improved rates of severe toxicity with the prophylactic use of dexamethasone (Oluwole OO, Br J Haematol 2021). There is limited data on which patients benefit the most from such strategies.
Methods: This is a study to evaluate factors associated with the development of severe immune mediated toxicities and treatment resistance in patients with R/R DLBCL treated with standard of care axicabtagene ciloleucel (axi-cel) at Moffitt Cancer Center. Baseline samples of CRP, LDH and ferritin were collected within one week prior to lymphodepleting chemotherapy. Cytokine analyses were performed as previously described (Faramand R, CCR 2020). Cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) were graded based on ASTCT criteria.
Patients were stratified into three risk groups based on baseline CRP and ferritin; low risk (CRP <4gm/dL and ferritin <400ng/dL), intermediate (not meeting criteria for high risk or low risk), and high risk (CRP >4gm/dL and ferritin >400ng/dL). During the study period, institutional clinical standards were revised to administer prophylactic corticosteroids consecutively to all patients meeting high risk criteria. Patient survival outcomes were compared using Kaplan-Meier curves and subsequent log-rank tests. Multivariable Cox proportional hazards regression model with adjustment for clinical covariates was used to estimate hazard ratios for survival outcomes.
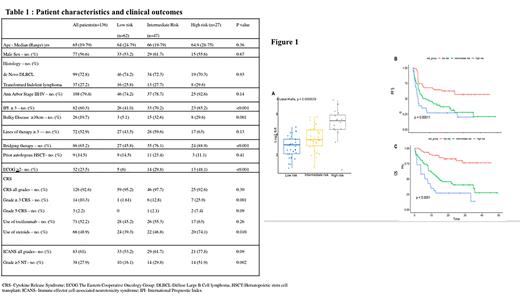
Results: 136 patients with R/R DLBCL treated with axi-cel (>2 lines of therapy) are included in this study. Baseline patient characteristics and outcomes are summarized in Table 1. Most patients developed CRS (n=126) with 10.3% developing severe (gr. >3) CRS. Eighty-three patients (61%) developed ICANS with 28% having severe gr. >3 toxicity. The median overall survival (OS) was 34.9 months with a median progression free survival (PFS) of 12.2 months.
We previously reported association of baseline elevated IL6 levels with poor clinical outcomes. Baseline IL6 levels correlated (p<0.0001, Fig. 1A) with risk category based on CRP and ferritin. Rates of severe CRS (25.9 vs. 1.61%, p=0.001) and ICANS ( 51.9 vs. 16.1%, p=0.002) were significantly higher in the high-risk (HR) group in comparison to the low risk group. Median PFS was 3 months, 7.9 and not reached in the high, intermediate and low risk groups respectively (p=0.0001) (Fig. 1B). The median OS was significantly inferior in the HR group at 6.9 months compared to 14.9 months and not reached in the intermediate and low groups respectively (p<0.0001) (Fig. 1C).
Based on the above risk stratification, ten additional patients met HR criteria and were treated with prophylactic dexamethasone for three days starting on the day of CAR T cell infusion. Although most patients (90%) developed CRS, none developed severe CRS in the prophylactic steroids group which compares favorably to 25.9% observed in historical HR patients who did not receive prophylactic steroids (p=0.16). Severe ICANS was lower (30%) in the prophylactic cohort as compared to 52% in historical HR patients (p=0.29). The median PFS and OS were 2.9 and 5.9 months respectively in recipients of prophylactic steroids.
Discussion: We establish that baseline elevated levels of CRP and Ferritin are associated with severe toxicity, and significantly inferior PFS and OS. Patients in the low risk group have excellent outcomes in terms of toxicity and efficacy using standard treatment paradigms negating the need for steroid prophylaxis and may be considered for outpatient treatment. In this report, prophylactic steroids were only used in 10 patients meeting high risk criteria. While there were no cases of severe CRS, efficacy outcomes remained poor in this high-risk cohort despite steroid prophylaxis. High risk patients can be easily identified prior to treatment with CAR T using standard labs and are encouraged to enroll on clinical trials to improve outcomes.
Disclosures
Faramand:Novartis: Research Funding; Kite/Gilead: Research Funding. Jain:Incyte: Research Funding; MyeloidTx: Consultancy; BMS: Consultancy; Novartis: Consultancy; Kite Pharma: Consultancy, Research Funding. Chavez:Beigene: Honoraria; Astrazeneca: Research Funding, Speakers Bureau; MorphoSys/Incyte: Speakers Bureau; TG Therapeutics: Honoraria; ADC Therapeutics: Research Funding; GenMab: Consultancy; Abbvie: Consultancy; Epizyme: Honoraria, Speakers Bureau; Kite Pharma: Consultancy; Adicet: Consultancy; Janssen: Research Funding; Merck: Research Funding. Shah:Servier: Other: grants and investigator-initiated trials; PeproMene Bio: Other: Steering committee; Autolus: Consultancy; Century Therapeutics: Consultancy; Adaptive: Consultancy; Pharmacyclics: Consultancy; Beigene: Consultancy; Acrotech: Consultancy; Jazz: Consultancy, Other: grants and investigator-initiated trials; Precision Biosciences: Consultancy; Kite/Gilead: Consultancy, Other: grants and investigator-initiated trials; BMS/Celgene/Juno: Consultancy; Novartis: Consultancy; Pfizer: Consultancy; Amgen: Consultancy. Pinilla Ibarz:SecuraBio: Research Funding; AstraZeneca: Consultancy; AbbVie: Consultancy; Pharmacyclics: Consultancy; Janssen Pharmaceuticals: Consultancy. Lazaryan:Teladoc: Current equity holder in publicly-traded company; AmWel: Current equity holder in publicly-traded company; Sanofi: Consultancy; AvroBio: Consultancy; Humanigen: Consultancy. Speth:Kite, a Gilead company: Current Employment. Song:Kite, a Gilead Company: Current Employment, Patents & Royalties: Gilead; Gilead Sciences: Current holder of stock options in a privately-held company, Patents & Royalties. Mattie:Kite/Gilead: Current Employment. Locke:Aptitude Health: Other: Education or editorial role; BioPharma Communications CARE Education: Other: Education or editorial role; Clinical Care Options Oncology: Other: Education or editorial role; Society for Immunotherapy of Cancer: Other: Education or editorial role; Imedex: Other: Education or editorial role; ASH: Other: Education or editorial role; Moffitt Cancer Center: Patents & Royalties: several patents held by the institution in his name (unlicensed) in the field of cellular immunotherapy; Gerson Lehrman Group: Consultancy; Emerging Therapy Solutions: Consultancy; EcoR1: Consultancy; Cowen: Consultancy; Leukemia and Lymphoma Society: Research Funding; National Cancer Institute: Research Funding; BMS: Research Funding; BlueBird Bio: Research Funding; Novartis: Research Funding; Allogene: Research Funding; Kite Pharma: Research Funding; Umoja: Membership on an entity's Board of Directors or advisory committees; Wugen: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Sana: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Legend Biotech: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees; Kite Pharma: Membership on an entity's Board of Directors or advisory committees; Iovance: Membership on an entity's Board of Directors or advisory committees; GammaDelta Therapeutics: Membership on an entity's Board of Directors or advisory committees; Cellular Biomedicine Group: Membership on an entity's Board of Directors or advisory committees; Caribou: Membership on an entity's Board of Directors or advisory committees; Calibr: Membership on an entity's Board of Directors or advisory committees; BMS/Celgene: Membership on an entity's Board of Directors or advisory committees; Bluebird Bio: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Allogene: Membership on an entity's Board of Directors or advisory committees; A2: Membership on an entity's Board of Directors or advisory committees. Davila:Atara: Other: licensing fees, Patents & Royalties: WO2019165156, Research Funding; CRISPR: Other: licensing fees, Research Funding; Novartis: Research Funding; Synthekine: Consultancy; Kite: Research Funding; Adicet: Consultancy; Bellicum: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal